We're beginning to understand the 'tumor microenvironment' that makes cancer so resilient
The cellular processes that help tumors thrive in the body are a promising new target for cancer treatments
Scientific research has made remarkable breakthroughs in understanding the human brain and treating diseases that affect it, including epilepsy, Huntington's disease, and dementia. But some ailments continue to evade our best attempts. For glioblastoma, the aggressive form of brain cancer that was diagnosed in Senator John McCain, a recent study demonstrated that the way forward might be a detour from the conventional research path.
After their diagnosis, patients with glioblastoma typically survive about 15 months, a statistic that hasn’t changed significantly in decades. While researchers have made extraordinary leaps in understanding the disease, the options for treating it have not kept pace.
However, a study from the Case Western Reserve University School of Medicine and Cleveland Clinic cast light on a previously unknown set of genes that glioblastoma needs to survive in the brain. Unlike conventional chemotherapy treatments, which work by targeting fast-dividing cells, medical student Tyler E. Miller and his colleagues found that in glioblastoma, it may be more effective to target a cellular stress response instead.
Glioblastoma cells growing in vitro
Sabari J, Lax D, Connors D, Brotman I, Mindrebo E, Butler C, Entersz I, Jia D, Foty R
Miller took glioblastoma cells from human patients and studied them in two ways: in vitro (Latin for in glass), whereby the cells were grown on a dish, and in vivo (Latin for in life), by implanting the cells in the brains of mice to regrow whole tumors. In the next phase of the study, Miller performed a genetic screen; he tested several hundred genes in vitro and in vivo to see which ones the cells relied on to survive.
The results were exciting and unexpected. To survive in mice, the cancer cells depended on 57 genes that had no impact on their growth in a dish. Of those genes, 12 were tightly linked to how the cells respond to a stressful environment, such as a lack of nutrients or challenging growth conditions. One gene, called JMJD6, was investigated in more depth because of its ability to regulate the functioning of numerous other genes. The authors found that depleting cells of JMJD6 in vitro didn’t affect their ability to survive and grow. But when the same JMJD6-deficient cells were implanted in mice, the tumors grew considerably slower and the mice survived significantly longer.
What sets apart Miller’s study is not only the novel cancer dependencies he unravels, but also the route he took to discover them. Traditionally, scientists begin their experiments in vitro because of the ease, speed, and lower cost of manipulating cells in a dish. With a sound theoretical framework and data to back us up, we then formulate a hypothesis to predict what would happen in vivo and begin performing experiments on mice or other organisms. Only then can we start to appreciate what is becoming increasingly important to the cancer research sphere: the tumor microenvironment.
Underneath the surface, cancerous tumors are teeming with cellular processes that support and bolster their own growth while shifting blood and nutrients away from nearby healthy cells. To the extent that tumors draw resources in from their environment, they also shape it, making it more hostile towards the body’s immune system and healthy cells. The result is a complex and seemingly endless number of chemical and physical interactions between tumor cells and the microenvironment around them, each side influencing the other.
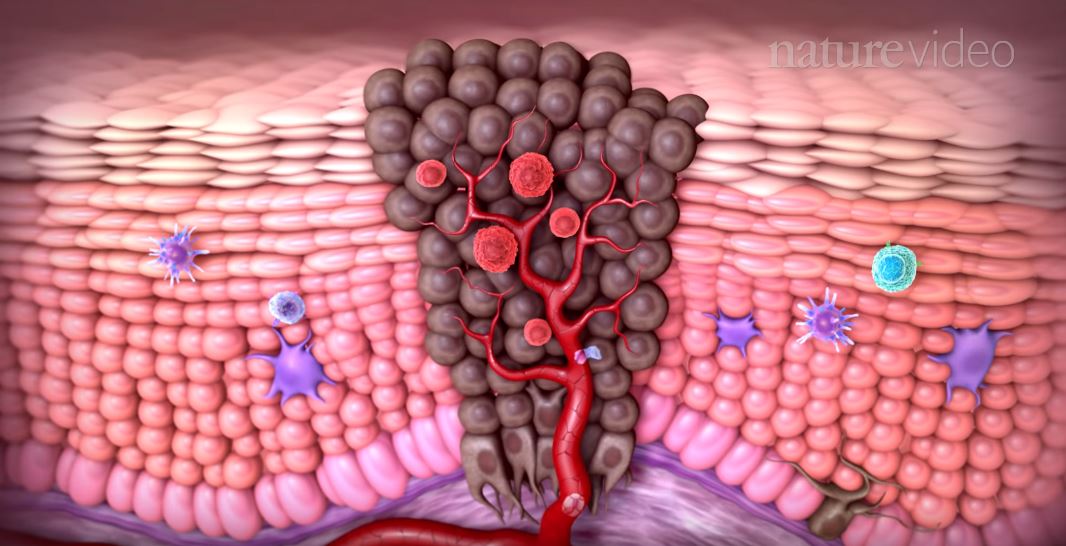
A rendering of a tumor microenvironment
Scientists who perform experiments in vitro know that the results are an imperfect predictor of what would happen in the dynamic in vivo environment. Indeed, there are countless examples of scientists whose groundbreaking findings in cells, and even in mice, were met with saddening results when they finally found their way to clinical studies. Despite meeting the rigorous demands of in vitro testing, even the most robust hypotheses can be fatally discounted by the complexities of experimenting in vivo.
Bearing this in mind, Miller’s decision to test both at once, and the ramifications that has for glioblastoma science, proves to be all the more remarkable.
As neuroscientist Jeremy Rich, a senior author in the study now at UC-San Diego, stated in a news release, “This means the field has been missing a number of potential therapeutic targets that may actually improve patient outcomes and prolong survival.” While the results of this study will surely trickle further down the research pipeline, Miller has also paved the way for future research to push for this novel approach to therapeutic discovery.
Rather than promoting the traditional perspective, this study provokes the question: what other novel aspects of cancer biology can we learn about by looking in vivo? All evidence points to the cellular mechanisms that guard tumor survival and sustain its growth in ways that we can’t comprehend in a dish.