To stop pancreatic cancer from spreading, cut out the chatter
By switching off cell-to-cell communication, researchers turned cancer’s bazooka into a rubber pistol
Photo by Kristina Flour on Unsplash
The pancreas is a multi-tasker. Unfortunately, so is its most common cancer.
A pancreatic cancer diagnosis can carry the weight of a death sentence – more than nine out of every 10 people don’t live to see the 5 year anniversary of their diagnosis. Celebrity diagnoses, like Alex Trebek's, popularize an anxiety felt by the roughly 50,000 people diagnosed each year. The disease progresses so quickly, and frequently without detection, that treatment is often futile. But, new research reveals how pancreatic cancer cells spread with ease – and how we might stop their hostile takeover.

The multitasking pancreas.
Shaped like a comma and nestled atop the small intestine, the pancreas is responsible for a vital arsenal of hormones that help maintain blood sugar levels. But it also spends time working with its downstairs neighbor, the intestine, to help us digest. It produces a thick “juice,” (actual technical term) that pours down a duct to supply the small intestine with enzymes that break down carbs, fats, and proteins. Cancer of that duct, pancreatic ductal adenocarcinoma, claims over 40,000 lives every year. By the time a doctor breaks the news to their patient, that cancer has likely spread elsewhere and will be difficult to treat.
Most cancers are riddled with avenues of perforated blood vessels. Ambitious cancer cells can use them to spread tumors elsewhere, but those avenues also make it easier to treat the disease with drugs. With this type of pancreatic duct cancer, it’s the worst of both worlds.
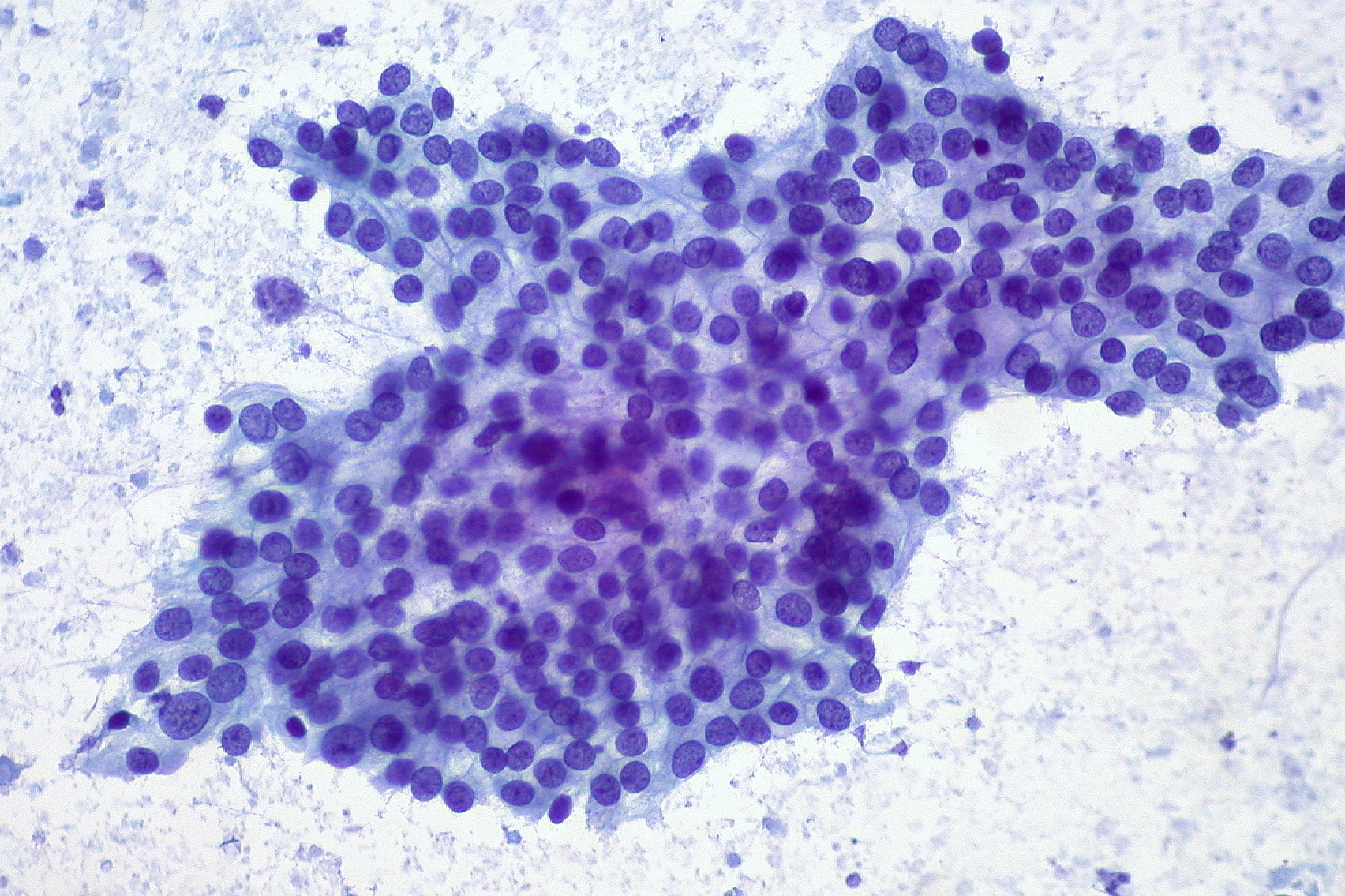
Adenocarcinoma of the pancreas. The dark purple circles in the center of the tumor are the nuclei of cancer cells.
Ed Uthman on Flickr
“They escape really early but they don’t have a lot of vessels,” says Duc-Huy Nguyen, a co-author of the study. A healthy pancreas has far more blood vessels than a cancerous one. According to Nguyen, some doctors have reported diseased pancreases with structures that look like normal blood vessels, but are in fact nonfunctional tubes lined with cancer cells rather than healthy ones. Nobody knew why or how the cancer could kill and replace healthy cells.
Nguyen knew that the answer likely had to do with a biological competition between different types of cells: cancerous pancreatic cells, and healthy endothelial cells that line nearby bloodstreams.
“You have two different cell types and you place them next to one another,” says Nguyen, “If you ask ‘who is going to win?’ I’d believe the tumor cell is going to.”
Nguyen and his team designed a clear plastic chip to support pancreatic cancer cells beside an artificial bloodstream lined with endothelial cells. With total visibility and control, the team could watch cancer cells inch towards their rivals. Nguyen expected to see a typical process called intravasation – when cancer cells casually slip past healthy ones to enter the bloodstream. He was surprised to witness something far more destructive.
_(cropped).jpg)
Alex Trebek's recent diagnosis of pancreatic cancer brought the condition into the limelight
Anders Krusberg/Peabody Awards via Wikimedia Commons
The cancer cells muscle their way into a stronghold over their neighbors. First, they sneak up on the endothelial cells; then they wrap around them like sleeve. Endothelial cells represent a sort of blockade, preventing undesired cells from entering the bloodstream. But this mob of cancer cells is aggressive, and quick to overpower this blockade meant to keep them out of the blood. Before long, this protective barrier of endothelial cells is no more, and cancerous cells fill the channel. The cancer can then juggle a lethal combination of offensive and defensive maneuvers: hijacking blood vessels to isolate itself from attack, and dispatching even more cancer cells to spread elsewhere.
Zooming in on the drama was interesting to Nguyen, but the science could leap from interesting to useful if he could devise a way to slow the cancer's progression. “So we just asked the question: what really happens there?” Nguyen and his team set out to eavesdrop on the chemical conversations going on between cells.
They began by tinkering with the cancer’s main line of communication with the outside world: a horde of chemicals and receptors called the transforming growth factor beta (TGF-β) signaling pathway. Finding the right chemical signal that prompts the cancer's outward progression, Nguyen says, could reveal a treatment that stops the takeover in its tracks.
The team quickly confirmed they were on to something. Using a TGF-β-inhibiting drug in their the chip-model, Nguyen switched off large chunks of the signaling pathway as soon as the cancer reached healthy cells. That change significantly reduced the blood vessel destruction.
Encouraged, the team used CRISPR gene editing to delete individual receptors in both cell types. One by one, Nguyen examined the cancer’s progress, hoping to pinpoint a secret weapon. He finally found that weapon in the form of a receptor called ALK7. By knocking out the pancreatic cancer cell’s ALK7, they rescued the endothelial cells from certain defeat. The team then repeated the experiment using mice carrying the genetically modified cancer and found that mouse blood vessels survived far better than those exposed to the unmodified cancer. Hampering that receptor amounted to swapping out the cancer’s bazooka for a rubber pistol.
“I thought this was important work,” says Melissa Skala, a cancer researcher at the Morgridge Institute not affiliated with the study. “There are very few treatment options for pancreatic cancer, so any development that enables more targeted drugs is very important.”
“I felt they were actually very careful with their claims,” says Shannon Stott, a scientist at Massachusetts General Hospital unaffiliated with this study. Stott creates devices to study how cancer spreads and paid keen attention to the design of the cancer model. She says this experiment has an important balance: it is simple enough to study the disease reliably, and complex enough to obtain potentially critical for real patients. “It’s a clever, elegant study.”
Since cancer is such a diverse disease, this treatment can’t treat every type of cancer. But Nguyen believes that some, such as ovarian cancer, may respond to the same treatment.
We’re still far from using this work to treat patients. Still, models like this one give us a platform to understand diseases and pinpoint their weaknesses. “My takeaway was not, ‘Oh, we can start treating patients with this’,” says Stott. “It opens the door for us to expand upon this to ask interesting questions.”
Peer Commentary
Feedback and follow-up from other members of our community
Kevin Pels
Chemical Biology
Dana-Farber Cancer Institute
Max describes the early-stage metastatic process of intravasation before introducing the research- a cool study that elucidates the molecular mechanism of how pancreatic cancer cells ablate healthy endothelial cells and take over blood vessels. However I keep going back to what Max said at the beginning of his article: often, by the time pancreatic cancer is diagnosed, it has already breached the vasculature and metastasized. Thus any future therapy evolving from this finding will be helpful in the minority of pancreatic cancer diagnoses occurring at an early stage, but its impact could be increased by further advances in detection of pancreatic cancer via genetic and/or CT-guided screening.
That’s a great point and it was echoed by the scientists I spoke with. I’m personally curious if someone can show the same metastatic invasion mechanism in a different cancer model – perhaps treatment would be more successful there.
Thanks for reading Kevin!